Warranty Policy
Scope
Ziacom Implants SLU, hereinafter “ZIACOM®”, guarantees the quality of the materials, manufacturing and proper functioning of its original ZIACOM® branded products, hereinafter the “ZIACOM® Product(s)”. The ZIACOM® Guarantee Programme, hereinafter the “Guarantee Programme”, is applicable to ZIACOM® Products. If a product is faulty, before or after its use, due to a failure in the resistance of the material and/or stability of the ZIACOM® Product during the period covered by the Guarantee Programme indicated in Section 3, notify this to ZIACOM® immediately following the Incident Notification procedure set out in Section 6 of this Guarantee Programme.
This Guarantee Programme refers exclusively to the ZIACOM® Products indicated in Section 3 and to the clinical or medical professional responsible for the treatment, hereinafter the “User”. The processing of any incident regarding a ZIACOM® Product must be managed solely and exclusively through this Guarantee Programme.
By purchasing any ZIACOM® Product and participating in this Guarantee Programme, the User accepts the terms and conditions set forth herein.
Beneficiaries
This Guarantee Programme is exclusively for the User and does not result in any type of liability vis-a-vis third parties, particularly patients or providers of any kind, who are not included in this guarantee. The coverage provided by the Guarantee Programme ONLY contemplates the substitution or replacement of the ZIACOM® Products for the same products included in the guarantee claim or for other equivalent products if the original ones are discontinued. Therefore, this Guarantee Programme does not cover any associated cost that may arise for other reasons.
Products covered
The Guarantee Programme covers the following products for the time described in the table below. The guarantee in this Programme does not exclude guarantees that the User may have as a consequence of their individual agreements with ZIACOM®.
| Implants | Lifetime guarantee. Replacement for an implant of the same type or equivalent 1 |
| Prosthetic components | 15-year guarantee. Replacement for a prosthetic component of the same type or equivalent 2 |
Important
Consumable components or products, reused single-use products and those that have a retention function are excluded from this Guarantee Programme.
- If this product has been discontinued or is no longer available on the market, it will be replaced by the product that has succeeded it or another equivalent within the ZIACOM® implants range.
- If this product has been discontinued or is no longer available on the market, it will be replaced by the product that has succeeded it or another equivalent within the ZIACOM® Prosthetic components range.
General terms and conditions
ZIACOM® will replace the faulty product(s) covered by this Guarantee Programme for another identical or equivalent product that has succeeded it if it has now been discontinued.
The guarantee periods described above will begin from the moment that the ZIACOM® Products are sold to the User. As a result, the start date of the Guarantee Programme for these products will be that appearing on the User’s sale invoice.
The Guarantee Programme will be valid so long as all the conditions of the Programme described below are fulfilled and reliably documented:
That the user
- Has directly purchased the ZIACOM® Product and this product has been purchased through an official ZIACOM® sales channel.
- Has selected the right model and type of implant for the surgical technique used and the planned treatment with implants.
- Has perfectly documented the patient’s consent.
- Has exclusively used ZIACOM® Products throughout the treatment and under no circumstance have these been modified or combined with elements or components from other manufacturers.
- Has performed and documented all regular check-ups established for the treatment and on the scheduled dates, verifying that the patient has good oral hygiene.
- Has consulted and complied with all protocols, work procedures, usage instructions for the ZIACOM® Products used during the treatment, both surgical and prosthodontic, and that were in force at the time of performing the treatment.
- Has properly selected and applied the appropriate and clinically accepted dental techniques for performing treatments with dental implant before, during and after treatment.
- Has reported and completed all mandatory parts of the ZIACOM® Guarantee Programme (Form-101) form within a maximum period of 30 calendar days after the occurrence of the incident that led to the use of the Guarantee Programme, providing all documentation requested by ZIACOM® for its investigation.
- Has not exceeded the annual 3% replacement rate for their past purchases. Only in the case of non-osseointegrated implants.
- Is up to date with all their contractual obligations with Ziacom Medical SL or any of its subsidiaries or associates at the time of requesting the Guarantee Programme.
- Has sent back to ZIACOM® the ZIACOM® Products that they want to return under the Guarantee Programme, duly packaged and sterilised, together with the documentation required to be able to process and study the guarantee. See conditions for returning products at the end of the document.
That the patient
- Does not have any of the contraindications described in the usage instructions for the ZIACOM® Product in force at the beginning, during and after the treatment.
- Has visited the User and performed all the regular check-ups established for the treatment and on the dates designated for this purpose.
- Has followed the control guidelines described by the User regarding the maintenance, care and good oral hygiene of the treatment with dental implants.
- That the patient has not manipulated, in whole or in part, by themselves or through third parties, the dental implant treatment performed.
The usage instructions, protocols and work and maintenance procedures for the ZIACOM® Products are described in the surgical and prosthodontic brochures and user manuals that accompany the ZIACOM® Products, or that can be consulted/downloaded directly from our website www.ziacom.com
Exclusion or Grounds for invalidation
Excluded from the Guarantee Programme are ZIACOM® Products that:
- Have not been used in accordance with the conditions of use and protocols specified by ZIACOM® as manufacturer and/or have been used in conjunction with products from other brands or manufacturers.
- Have undergone any type of contamination by the User or third parties unrelated to ZIACOM®, its network of subsidiaries or authorised official distributors.
- Have been handled and/or applied incorrectly, in whole or in part, in any phases of the treatment or have been handled and/or applied by staff members who are not qualified or do not have the qualifications needed to do this according to the legislation in force in each country.
- Have reused single-use ZIACOM® Products.
- Have not complied with any of the points listed in Section 4 on general conditions of the ZIACOM® Guarantee Programme.
Any damage caused to ZIACOM® Products as a result of or arising from, in whole or in part, trauma, accidents or any other damage caused by the patient themselves or by a third party, is completely excluded from the Guarantee Programme.
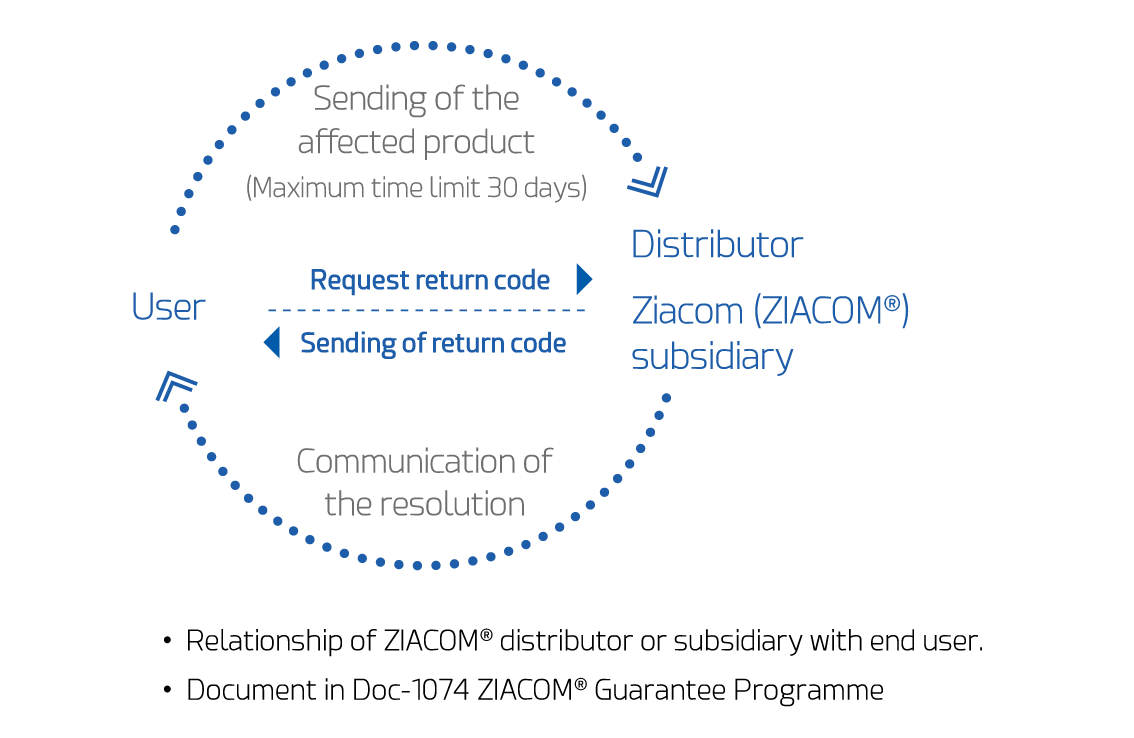
Notification of incidents and request
To request the guarantee under the Guarantee Programme due to an incident with a product, you must report this within a maximum of 30 calendar days after it occurs, following the procedure detailed below:
- Send the guarantee programme application form fully completed with all required information and any extra information you can provide to explain the case or circumstances.
- Send the product you wish to have replaced under the Guarantee Programme packaged in a sterilisation bag and properly sterilised; together with the form mentioned in the previous point. If the product is not sent packaged in a sterilisation bag and sterilised in an autoclave (see the cleaning, disinfection and sterilisation instructions on the website), the Quality Department will return it to the clinic. The Customer Service Department at ZIACOM® or its authorised official distributor will provide you with a personalised Guarantee Programme Code, sent by e-mail, together with the corresponding application form (Form-101) that must be completed and sent according to the instructions for accessing the Guarantee Programme. This form can also be downloaded from the website ziacom.com
- The ZIACOM® Quality Department will not start studying any incident under guarantee until it has received all documentation, perfectly completed, and the product(s) covered by this guarantee. The Quality Department will request the information required to start the investigation. In the event that the form is not completed correctly, the Quality Department will return the form and the product to the clinic.
- The analysis of the documentation by the Quality Department at ZIACOM® will take place within a maximum period of 30 calendar days from all documentation required for the study arriving at the ZIACOM® regional headquarters. After that time, the Quality Department will issue a decision in writing to the User with the resolution of the Guarantee Programme, in which it will accept or reject the request for this guarantee.
- In the event that the Quality Department does not receive the form correctly completed, the product correctly sterilised and the information required to study the case within a period of 30 days from the initial request, the Quality Department may reject the Guarantee Programme, making it impossible for the customer to make a claim.
If the maximum period of 30 calendar days expires without our Quality Department receiving all documentation required to analyse this guarantee, the Quality Department at ZIACOM® will archive this file as “Not applicable or rejected due to lack of data”. It will not be possible to reopen the file and the right to this guarantee will be lost. *The sterilisation must be performed exclusively using the ZIACOM® sterilisation protocol (Document Doc-1066). Note. The implants to be returned must have been packaged and sterilised according to the aforementioned procedure, but never cleaned.
Limits and limitations
- Only ZIACOM®, as manufacturer of its products, can specify the guarantee conditions of its ZIACOM® Products, so no company or individual who works with, markets or distributes ZIACOM® Products can define guarantee conditions different to those indicated in this Guarantee Programme.
- This Guarantee Programme is the only guarantee provided by ZIACOM® for its products and must be applied in addition to the guarantees that the User may have as a consequence of their individual agreements with ZIACOM®.
- ZIACOM® rejects any other type of guarantee or liability, express or implied, whole or partial, with respect to the User for any loss of profits, lost earnings, special damages, direct or indirect, unforeseen or consequential, of any nature and due to any cause, as well as collateral or consequential damages directly or indirectly related to ZIACOM® Products, services or information.
- The use and application of ZIACOM® Products is beyond the manufacturer’s control, remaining the sole responsibility of the User of the ZIACOM® Products. All damages that the User could cause to themselves or to third parties derived from the use of the ZIACOM® Products is their responsibility and ZIACOM® and all of its subsidiaries and/or authorised official distributors are exempt from liability for damages or losses derived from the mishandling or misuse of the ZIACOM® Products. The Guarantee Programme only applies to the ZIACOM® Products manufactured by Ziacom Medical, S.L. and marketed through the means mentioned above.
- By purchasing any ZIACOM® Product and participating in this Guarantee Programme, the User accepts the terms and conditions set forth herein.
Geographical limitation
This Guarantee Programme is only valid for ZIACOM® Products sold in any country where ZIACOM®, any of its subsidiaries or an official distributor authorised by ZIACOM® is present and has an office.
Modification, cancellation or termination
Ziacom Implants SLU may modify, cancel or terminate, in whole or in part, and at any time, this Guarantee Programme, so long as Ziacom Implants SLU notifies the changes made to its Guarantee Programme in writing at least 60 calendar days before they come into effect. The disclosure about these changes will be deemed fully valid and notified if this disclosure is made through its publication on the website www.ziacom.com. Therefore, we recommend regularly consulting the updated version of the Guarantee Programme.
Any modifications, cancellations or the termination of the Guarantee Programme will not affect the ZIACOM® Products subject to this Guarantee Programme up to that moment and reported to ZIACOM® prior to the modification, cancellation or termination date.
Final provisions
Ziacom Implants SLU reserves the right to terminate commercial relations with the User, the company in which they participate, the company at which they work or the entity they represent that, due to a repeated breach of protocols, surgical or prosthodontic procedures or due to not following the guidelines set in the usage instructions for the ZIACOM® Products, gives rise to partial and unfounded negative opinions, directly disclosed or published, and causes harm to the ZIACOM® brand or Ziacom Implants SLU.
Ziacom Implants SLU reserves the right to take any and all legal actions to which it is entitled in order to defend its image rights and to preserve the brand image and the good reputation of its products in the market.
Ziacom Implants SLU- Spain
Att. Departamento de Calidad Programa de Garantía ZIACOM®
Calle Búhos, 2
28320 Pinto - Madrid - ESPAÑA
Phone: +34 91 723 33 06
Email: info@ziacom.es
Ziacom Medical USA LLC – USA
Miami – EEUU
333 S.E 2nd Avenue, Suite 2000
Miami, FL 33131 -USA
Phone: +1(786) 224 – 0089
Email: info.usa@ziacom.es
Ziacom Lusobionic Portugal Lda - Portugal
Avenida Miguel Bombarda , 36 – 5 B
1050-165 Lisboa – Portugal
Phone: +351 215850209
Email: info@ziacom.pt
Ziacom Safe Implant - Ecuador
Av. iñaquito, edificio Metropolitan, Oficina 304
170155 – Quito
Phone: +593 99 368 0879
Email: jcampoverde@safeimplant.com.ec
Oak Business Sas - Colombia
Diagonal 71b, Nº 98-04. Planta 1 (Alamos Norte)
111141 - Bogota
Teléfono +57 300 266 7624
Contact name: David Alexander Bernal Suarez
Email: david.suarez@oakbusiness.com.co
Sas Eck France - France
97 Allée De Chantoiseau (3eme Maison a Gauche)
38330 - Saint Ismier
Teléfono:+33766076262
Contact name: Julien Luis André Lebreux
TopDent Company - Egypt
Nasser City, Cairo
Phone: 0020 102 700 0808
Contact name: Dr. Ahmed Hashem
Email: dentist.hashem88@gmail.com
Ziacom ITS
Viale del Lavoro 14
35010 Vigonza (PD)
Teléfono:+39 328 693 6816
Email: info.shop@ziacom.it info@coemf.it
Biovico Sp. Z O.O. - Poland
Hutnicza 15B
81-061 Gdynia
Phone: +48 572 570 292
Contact name: Natalia Blewąska
Email: natalia.blewaska@biovico.com
On Dental Pazarlama Ticaret Ltd.Sti – Turkey
Mesrutiyet Mh. Şair Nigar Sokak, Aycibin Business Center 6/25
34363 – Sisli – Istanbul
Phone: +90 5326586056
Contact name: Kutay Eryılmaz
Email: kutay@ondental.com.tr
Manh Phu Dental Distribution Company Limited - Vietnam
1112/9A Kha Van Can Street, Linh Chieu Ward, Thu Duc City
HCM City, Viet Nam
Phone: +84 283 636 52 22
Contact name: Nguyễn Tiến Phú
Email: info.manhphu@gmail.com
Mena Biotech Facilities Management - Jordan, United Arab Emirates, Kuwait, Saudi Arabia, Qatar e Iraq
Empire Heights, Business Bay,
Dubai – UAE
Phone: +965 6066 6670
Contact name: Mr. Fadi Odeh
Email: fadiodeh@menabiotech.com
Niltorg LLC – Russia, Belarus, Kazakhastan y Uzbekistan
Street Elektrodnaya 2 Pomishenie VII/3
111524 Moscú
Phone: +7 985 468-04-90
Contact name: Mr Nilesh Joshi
Email: info@niltorg.com
PE Niltolrg Ukraine - Ukraine
Kharkivske Shosse, Building 19,
02090 Kyiv city
Phone: +380 50 312 0051
Contact name: Mr. Alexander Chupryna
Email: avantis.alexandr@gmail.com
Consult the updated list of ZIACOM® subsidiaries and authorised official distributors on the website


